26 August 2021
Not everyone responds the same way to a vaccine. Safeguarding our community from infectious diseases like COVID-19 means ensuring everyone is equally protected.
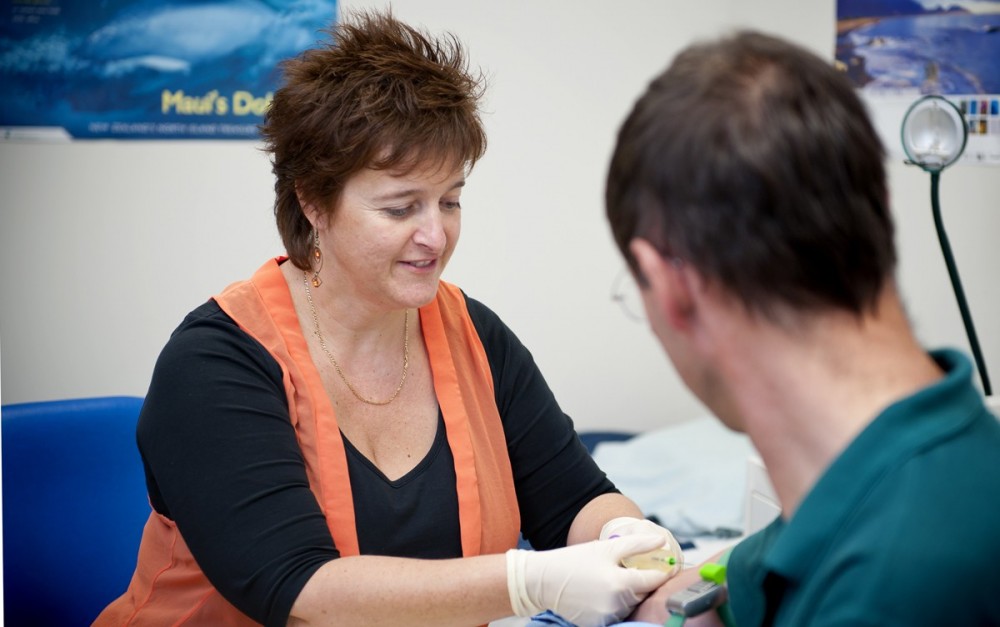
In early June, Vaccine Alliance Aotearoa New Zealand – Ohu Kaupare Huaketo (VAANZ) launched a COVID-19 clinical study at two sites in Rotorua and Christchurch to provide valuable information on how our uniquely ‘Kiwi’ population responds to the Pfizer-BioNTech COVID-19 vaccine.
The study, ‘Ka Mātau, Ka Ora’ (from knowledge comes wellbeing), aims to track and monitor participants’ immune responses to the vaccine over the course of a year in order to inform the national COVID-19 strategy and ultimately enhance vaccine effectiveness and confidence in New Zealand.
“We want to confirm if the immune response in New Zealand is what is seen internationally,” says the Malaghan Institute’s Dr Fran Priddy, VAANZ Clinical Director.
“While the Pfizer-BioNTech vaccine has demonstrated efficacy and safety in pivotal clinical trials and real-world studies, it has not yet been studied in New Zealand,” says Dr Priddy. “That’s not to imply that we’re at all worried that the vaccine is not going to be effective in populations here. Rather, we want to understand how New Zealanders’ immune systems respond to the vaccine, particularly in populations likely at higher risk from COVID-19, such as Māori, Pasifika and the elderly.”
Dr Priddy says with vaccine safety already being closely monitored and evaluated in New Zealand and internationally, Ka Mātau, Ka Ora will focus on characterising immune responses.

“Studies done post-vaccination in other countries have shown lower antibody responses in some groups, such as the elderly and those with obesity.
“We don’t know if this translates to reduced effectiveness, but it will be very hard to measure effectiveness unless we have a large outbreak. So measuring immune responses is the best proxy right now for us in New Zealand.
“I think the data from our study is likely to be confirmatory and supportive, and should make people feel more confident that the vaccine is going to work across Kiwi populations”.
Dr Priddy says our ‘COVID-naïve’ population will also offer unique data to global research. “Vaccine immune responses may differ in populations with little prior viral exposure, as is currently the situation in New Zealand.”
There are ways to increase immune responses to vaccines says Dr Priddy, so if the study identifies responses that may impact effectiveness, this could be addressed in the future by booster shots, different vaccination schedules, or different vaccine types.
Clinical immunologist Dr Maia Brewerton, who is a member of the Institute’s Trust Board and Te Urungi Māori advisory board, says that Aotearoa is in an enviable position due to our elimination strategy and measuring specific immune markers offers a useful alternative approach to assess the vaccine response amongst our people.
“Māori and Pasifika have a greater burden of conditions like heart disease associated with more severe COVID-19 disease, however even after we account for these conditions, Māori and Pasifika are still at increased risk of developing severe disease. We know there is more to learn and this research is important to help identify and understand any differences in the immune response which can guide the optimal vaccine approach for our people,” she says.
“During the influenza pandemics last century, Māori experienced higher death rates and I hope with knowledge from research like this we can prevent a repeat of this story for Māori as we journey into the uncertain future of this COVID-19 pandemic together – Ka Mātau, Ka Ora.”
The Malaghan Institute is running Ka Mātau, Ka Ora in conjunction with Lakeland Clinical Trials in Rotorua, and Southern Clinical Trials in Christchurch.
Related articles

In Focus: Rejuvenating the ageing immune system
17 July 2024

Kia Niwha Leader Fellowship for Malaghan vaccine researcher
6 May 2024

In Focus: Mapping the lung's fight – how the entire organ responds to infection
18 April 2024

Malaghan and National Institutes of Health research receives prestigious award
5 March 2024

In Focus: Tailoring mRNA vaccines for immunocompromised populations
14 December 2023
Scientists identify interferon-gamma as potential SARS-CoV-2 antiviral
13 December 2023
