Dr Robert Weinkove is Clinical Director and leads the Weinkove Laboratory's CAR T-cell programme. His research interests include cancer immunotherapy, B-cell malignancies, and haemato-oncology supportive care.
He also practices as a Consultant Haematologist at Te Rerenga Ora Blood & Cancer Centre and Kenepuru Hospital, Porirua.
Rob studied medicine at the University of Cambridge and Kings College London, trained in General Medicine and Haematology at Guy's and St Thomas' Hospitals in London and the Medizinische Hochschule Hannover in Germany, and completed an Immunology PhD with the University of Otago.
MA (Cantab), MBBS (Hons), PhD, FRACP, FRCP
Related news
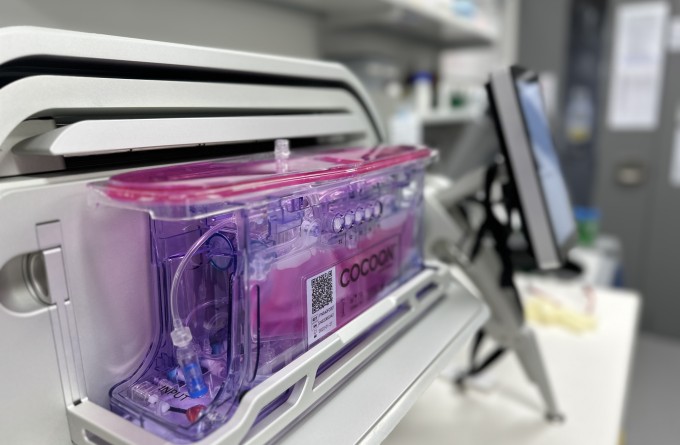
Significant milestone reached in first NZ CAR T-cell trial as preparations made for larger phase 2 registration trial
25 March 2024
Third-generation anti-CD19 CAR T-cells demonstrate efficacy without neurotoxicity in B-cell lymphoma phase 1 clinical trial
12 December 2023

Trial results offer hope to Kiwis with incurable blood cancer
3 November 2023

BioOra appoints CAR T-cell therapy pioneer, Professor Carl June to Board
31 October 2023

Malaghan Institute and BioOra deliver automated manufacturing to scale up CAR T-cell cancer therapy in NZ
16 August 2023

RNZ: New Zealand developed cancer treatment one step closer to market
17 May 2023
Publications
2023
Campbell A, Teh B, Mulligan S, Ross DM, Weinkove R, Gilroy N, Gangatharan S, Prince HM, Szer J, Trotman J, Lane S, Dickinson M, Quach H, Enjeti AK, Ku M, Gregory G, Hapgood G, Ho PJ, Cochrane T, Cheah C, Greenwood M, Latimer M, Berkahn L, Wight J, Armytage T, Diamond P, Tam CS, Hamad N (2023). Australia and New Zealand consensus position statement: use of COVID-19 therapeutics in patients with haematological malignancies. Intern Med J. 2023 Dec 25
Buckstein R, Callum J, Prica A, Bowen D, Wells RA, Leber B, Heddle N, Chodirker L, Cheung M, Mozessohn L, Yee K, Gallagher J, Parmentier A, Jamula E, McQuilten Z, Wood EM, Weinkov R, Zhang L, Mamedov A, Stanworth SJ, Lin Y (2023). Red cell transfusion thresholds in outpatients with myelodysplastic syndromes: Combined results from two randomized controlled feasibility studies. Am J Hematol. 2023 Dec 21
Mo A, Weinkove R, Wood EM, Shortt J, Johnston A, McQuilten ZK; Australasian Leukaemia and Lymphoma Group Supportive Care Scientific Working Party (2023). Use of platelet transfusions and tranexamic acid in patients with myelodysplastic syndromes: A clinical practice survey. Eur J Haematol. 2023 Dec 20
Robichon K, Bibi R, Kiernan M, Denny L, Prisinzano TE, Kivell BM, La Flamme AC (2023). Enhanced and complementary benefits of a nalfurafine and fingolimod combination to treat immune-driven demyelination. Clin Transl Immunology. 12(12):e1480
Brown JR, Eichhorst B, Hillmen P, Jurczak W, Kaźmierczak M, Lamanna N, O'Brien SM, Tam CS, Qiu L, Zhou K, Simkovic M, Mayer J, Gillespie-Twardy A, Ferrajoli A, Ganly PS, Weinkove R, Grosicki S, Mital A, Robak T, Osterborg A, Yimer HA, Salmi T, Wang MD, Fu L, Li J, Wu K, Cohen A, Shadman M (2023). Plain language summary of zanubrutinib or ibrutinib in chronic lymphocytic leukemia that is resistant to treatment or has come back after treatment. Future Oncol. 2023 Dec 13
Kirman JR, Weinkove R, Borger JG (2023). Immunology across two islands: understanding the research landscape of Aotearoa (New Zealand). Immunol Cell Biol. 2023 Nov 3
Cochrane T, Enrico A, Gomez-Almaguer D, Hadjiev E, Lech-Maranda E, Masszi T, Nikitin E, Robak T, Weinkove R, Wu SJ, Manzoor BS, Busman T, Pai M, Komlosi V, Anderson MA (2023). Updated quality of life data from the phase 3b VENICE II trial: patients with relapsed or refractory chronic lymphocytic leukemia receiving venetoclax monotherapy. Leuk Lymphoma. 2023 Aug 25:1-4
Nouri, Y., Weinkove, R., & Perret, R. (2023). An in vitro model to assess CRS potential of CAR T cells using a tumor cell line and autologous monocytes. Current Protocols, 3, e864
Yuen HLA, Weinkove R, Ullman A, Marsh N, Rickard CM, Chunilal S, McQuilten Z (2023). Central venous access device practice across haematology and oncology centres in Australia and New Zealand: a cross-sectional survey. Intern Med J. 53(3):426-43
Dasyam N, Sharples KJ, Barrow C, Huang Y, Bauer E, Mester B, Wood CE, Authier-Hall A, Dzhelali M, Ostapowicz T, Kumar R, Lowe J, Maxwell A, Burn OK, Williams GM, Carley SE, Caygill G, Jones J, Chan STS, Hinder VA, Macapagal J, McCusker M, Weinkove R, Brimble MA, Painter GF, Findlay MP, Dunbar PR, Gasser O, Hermans IF (2023). A randomised controlled trial of long NY-ESO-1 peptide-pulsed autologous dendritic cells with or without alpha-galactosylceramide in high-risk melanoma. Cancer Immunol Immunother. 2023 Mar 7
2022
Brown JR, Eichhorst B, Hillmen P, Jurczak W, Kaźmierczak M, Lamanna N, O'Brien SM, Tam CS, Qiu L, Zhou K, Simkovic M, Mayer J, Gillespie-Twardy A, Ferrajoli A, Ganly PS, Weinkove R, Grosicki S, Mital A, Robak T, Osterborg A, Yimer HA, Salmi T, Wang MD, Fu L, Li J, Wu K, Cohen A, Shadman M (2022). Zanubrutinib or Ibrutinib in Relapsed or Refractory Chronic Lymphocytic Leukemia. N Engl J Med. 388(4):319-332
Hillmen P, Eichhorst B, Brown JR, Lamanna N, O'Brien SM, Tam CS, Qiu L, Kazmierczak M, Zhou K, Šimkovič M, Mayer J, Gillespie-Twardy A, Shadman M, Ferrajoli A, Ganly PS, Weinkove R, Grosicki S, Mital A, Robak T, Österborg A, Yimer HA, Salmi T, Ji M, Yecies J, Idoine A, Wu K, Huang J, Jurczak W (2022). Zanubrutinib Versus Ibrutinib in Relapsed/Refractory Chronic Lymphocytic Leukemia and Small Lymphocytic Lymphoma: Interim Analysis of a Randomized Phase III Trial. J Clin Oncol. 41(5):1035-1045
Mulligan SP, Opat S, Cheah CY, Kuss B, Hertzberg M, Marlton P, Poplar S, Puig A, McGeachie M, Weinkove R, Tam CS (2022). Real-world experience of Australian and New Zealand patients with chronic lymphocytic leukemia and mantle cell lymphoma accessing ibrutinib through a Named Patient Program. Leuk Lymphoma. 2022 Nov 24:1-7
Hillmen P, Eichhorst B, Brown JR, Lamanna N, O'Brien SM, Tam CS, Qiu L, Kazmierczak M, Zhou K, Šimkovič M, Mayer J, Gillespie-Twardy A, Shadman M, Ferrajoli A, Ganly PS, Weinkove R, Grosicki S, Mital A, Robak T, Österborg A, Yimer HA, Salmi T, Ji M, Yecies J, Idoine A, Wu K, Huang J, Jurczak W (2022). Zanubrutinib Versus Ibrutinib in Relapsed/Refractory Chronic Lymphocytic Leukemia and Small Lymphocytic Lymphoma: Interim Analysis of a Randomized Phase III Trial. J Clin Oncol. 2022 Nov 17
Perram J, Purtill D, Bajel A, Butler J, O'Brien T, The B, Gilroy N, Ho PJ, Doocey R, Hills T, Perera T, Douglas G, Ramachadran S, Chee L, Trotman J, Weinkove R, Keogh S, Fraser C, Cochrane T, Watson AM, Diamond P, Latimer M, Irving I, Blyth E, Cheah C, Cole T, Milliken S, Yang H, Greenwood M, Bardy P, Kennedy G, Larsen S, Conyers R, Hamad N (2022). ANZTCT Position Statement: COVID-19 Management in Haematopoietic Stem Cell Transplant and Chimeric Antigen Receptor T cell Patients. Intern Med J. 2022 Nov 13
Qin L, Cui Y, Yuan T, Chen D, Zhao R, Li S, Jiang Z, Wu Q, Long Y, Wang S, Tang Z, Pan H, Li X, Wei W, Yang J, Luo X, Zhang Z, Tang Q, Liu P, Weinkove R, Yao Y, Qin D, Thiery JP, Li P (2022). Co-expression of a PD-L1-specific chimeric switch receptor augments the efficacy and persistence of CAR T cells via the CD70-CD27 axis. Nat Commun. 13(1):6051
Brown JR, Hillmen P, Eichhorst B, Lamanna N, O'Brien S, Tam CS, Qiu L, Kazmierczak M, Zhou K, Šimkovič M, Mayer J, Gillespie-Twardy A, Shadman M, Ferrajoli A, Ganly PS, Weinkove R, Salmi T, Wu K, Novotny W, Jurczak W (2022). CLL-115 First Interim Analysis of ALPINE Study: Results of a Phase 3 Randomized Study of Zanubrutinib vs Ibrutinib in Patients With Relapsed/Refractory (R/R) Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (CLL/SLL). Clin Lymphoma Myeloma Leuk. 22 Suppl 2:S266
Chai KL, Wong JWK, Weinkove R, Keegan A, Crispin PJ, Stanworth SJ, Morrissey O, Wood EM, McQuilten Z (2022). Interventions to reduce infections in patients with hematological malignancies: a systematic review and meta-analysis. Blood Adv. 2022 Jul 26
Burn OK, Farrand K, Pritchard T, Draper S, Tang CW, Mooney AH, Schmidt AJ, Yang SH, Williams GM, Brimble MA, Kandasamy M, Marshall AJ, Clarke K, Painter GF, Hermans IF, Weinkove R (2022). Glycolipid-peptide conjugate vaccines elicit CD8+ T-cell responses and prevent breast cancer metastasis. Clin Transl Immunology. 11(7):e1401
Jiang Z, Qin L, Tang Y, Liao R, Shi J, He B, Li S, Zheng D, Cui Y, Wu Q, Long Y, Yao Y, Wei Z, Hong Q, Wu Y, Mai Y, Gou S, Li X, Weinkove R, Norton S, Luo W, Feng W, Zhou H, Liu Q, Chen J, Lai L, Chen X, Pei D, Graf T, Liu X, Li Y, Liu P, Zhang Z, Li P (2022). Human induced-T-to-natural killer cells have potent anti-tumour activities. Biomark Res. 10(1):13
2021
Fernando SS, Paige EK, Dendle C, Weinkove R, Kong DCM, Omond P, Routledge DJ, Szer J, Blyth CC; Australasian Antifungal Guidelines Steering Committee (2021). Consensus guidelines for improving patients' understanding of invasive fungal disease and related risk prevention in the haematology/oncology setting, 2021. Intern Med J. 51 Suppl 7:220-233
McQuilten ZK, Busija L, Seymour JF, Stanworth S, Wood EM, Kenealy M, Weinkove R; Australasian Leukaemia and Lymphoma Group (ALLG) (2021). Hemoglobin is a key determinant of quality of life before and during azacitidine-based therapy for myelodysplasia and low blast count acute myeloid leukemia. Leuk Lymphoma. 63(3):676-683
Nouri Y, Weinkove R, Perret R (2021). T-cell intrinsic Toll-like receptor signaling: implications for cancer immunotherapy and CAR T-cells. J Immunother Cancer. 9(11):e003065
Jarvis E-M, Collings S, Authier-Hall A, Dasyam N, Luey B, Nacey J, Painter GF, Delahunt B, Hermans IF and Weinkove R (2021). Mucosal-Associated Invariant T (MAIT) Cell Dysfunction and PD-1 Expression in Prostate Cancer: Implications for Immunotherapy. Front. Immunol. 12:748741
Cochrane T, Enrico A, Gomez-Almaguer D, Hadjiev E, Lech-Maranda E, Masszi T, Nikitin E, Robak T, Weinkove R, Wu SJ, Sail KR, Pesko J, Pai M, Komlosi V, Anderson MA (2021). Impact of venetoclax monotherapy on the quality of life of patients with relapsed or refractory chronic lymphocytic leukemia: results from the phase 3b VENICE II trial. Leuk Lymphoma. 1-11
Weinkove R, George P, Ruka M, Haira TH, Giunti G (2021). Chimeric antigen receptor T-cells in New Zealand: challenges and opportunities. N Z Med J. 134(1542):96-108
2020
Di Ciaccio P, McCaughan G, Trotman J, Ho PJ, Cheah CY, Gangatharan S, Wight J, Ku M, Quach H, Gasiorowski R, Polizzotto MN, Prince HM, Mulligan S, Tam CS, Gregory G, Hapgood G, Spencer A, Dickinson M, Latimer M, Johnston A, Armytage T, Lee C, Cochrane T, Berkhahn L, Weinkove R, Doocey R, Harrison SJ, Webber N, Lee HP, Chapman S, Campbell BA, Gibbs SDJ, Hamad N (2020). Australian and New Zealand consensus statement on the management of lymphoma, chronic lymphocytic leukaemia and myeloma during the COVID-19 pandemic. Intern Med J.
Weinkove R, McQuilten ZK, Adler J, Agar MR, Blyth E, Cheng AC, Conyers R, Haeusler GM, Hardie C, Jackson C, Lane SW, Middlemiss T, Mollee P, Mulligan SP, Ritchie D, Ruka M, Solomon B, Szer J, Thursky KA, Wood EM, Worth LJ, Yong MK, Slavin MA, Teh BW (2020). Managing haematology and oncology patients during the COVID-19 pandemic: interim consensus guidance. Med J Aust. 2020
George P, Brown A, Weinkove R (2020). B-cell Prolymphocytic Leukaemia With a t(4;14) FGFR3/IGH Translocation: Response to Ibrutinib. Pathology. 52(4):491-492.
Grasso C, Field CS, Tang CW, Ferguson PM, J Compton B, Anderson RJ, Painter GF, Weinkove R, F Hermans I, Berridge MV (2020). Vaccines adjuvanted with an NKT cell agonist induce effective T-cell responses in models of CNS lymphoma. Immunotherapy.
Dasyam N, George P, Weinkove R (2020). Chimeric antigen receptor T-cell therapies: Optimising the dose. Br J Clin Pharmacol.
George P, Dasyam N, Giunti G, Mester B, Bauer E, Andrews B, Perera T, Ostapowicz T, Frampton C, Li P, Ritchie D, Bollard CM, Hermans IF, Weinkove R (2020). Third-generation anti-CD19 chimeric antigen receptor T-cells incorporating a TLR2 domain for relapsed or refractory B-cell lymphoma: a phase I clinical trial protocol (ENABLE). BMJ Open.10(2):e034629
Stanworth SJ, Killick S, McQuilten ZK, Karakantza M, Weinkove R, Smethurst H, Pankhurst LA, Hodge RL, Hopkins V, Thomas HL, Deary AJ, Callum J, Lin Y, Wood EM, Buckstein R, Bowen D; REDDS Investigators (2020). Red cell transfusion in outpatients with myelodysplastic syndromes: a feasibility and exploratory randomised trial. Br J Haematol. 189(2):279-290
2019
Weng J, Lai P, Qin L, Lai Y, Jiang Z, Luo C, Huang X, Wu S, Shao D, Deng C, Huang L, Lu Z, Zhou M, Zeng L, Chen D, Wang Y, Chen X, Geng S, Weinkove R, Tang Z, He C, Li P, Du X (2019). Correction to: A novel generation 1928zT2 CAR T cells induce remission in extramedullary relapse of acute lymphoblastic leukemia. J Hematol Oncol. 12(1):117
Dickinson M, Weinkove R (2019). Maintaining a fit T-cell compartment: lymphoma treatment sequencing in the era of chimeric antigen receptor T-cell therapies. Intern Med J. 49(10):1338
Compton BJ, Farrand KJ, Tang CW, Osmond TL, Speir M, Authier-Hall A, Wang J, Ferguson PM, Chan STS, Anderson RJ, Cooney TR, Hayman CM, Williams GM, Brimble MA, Brooks CR, Yong LK, Metelitsa LS, Zajonc DM, Godfrey DI, Gasser O, Weinkove R, Painter GF, Hermans IF (2019) Enhancing T cell responses and tumour immunity by vaccination with peptides conjugated to a weak NKT cell agonist. Org & Biomol Chem 17(5):1225-1237
Wong J, Wood EM, Crispin P, Weinkove R, McQuilten ZK; Australasian Leukaemia and Lymphoma Group (ALLG) Supportive Care Group (2019) Managing hypogammaglobulinaemia secondary to haematological malignancies in Australia and New Zealand: a clinician survey. Intern Med J49(3):358-363
Fischer K, Al-Sawaf O, Bahlo J, Fink AM, Tandon M, Dixon M, Robrecht S, Warburton S, Humphrey K, Samoylova O, Liberati AM, Pinilla-Ibarz J, Opat S, Sivcheva L, Le Dû K, Fogliatto LM, Niemann CU, Weinkove R, Robinson S, Kipps TJ, Boettcher S, Tausch E, Humerickhouse R, Eichhorst B, Wendtner CM, Langerak AW, Kreuzer KA, Ritgen M, Goede V, Stilgenbauer S, Mobasher M, Hallek M (2019) Venetoclax and Obinutuzumab in Patients with CLL and Coexisting Conditions. N Engl J Med 380(23):2225-2236
Weinkove R, George P, Dasyam N, McLellan AD (2019) Selecting costimulatory domains for chimeric antigen receptors: functional and clinical considerations. Clin Transl Immunology 8(5):e1049
Weinkove R, Bowden E, Wood C, Campion V, Carter J, Hall R, Weatherall M, Beasley R, Young P (2019) A randomized controlled feasibility trial of paracetamol during febrile neutropenia in hemato-oncology patients. Leukemia & Lymphoma 1-8 60(6):1540-1547
2018
Wheeler M, White G, Brockie S, Dickson M, Weinkove R (2018) Flow Cytometric Analysis of Mechanically Disaggregated Bone Marrow Trephine Biopsies. Cytometry B Clin Cytom94(6):935-940
Weng J, Peilong L, Le Q, Yunxin L, Zhiwu J, Chenwei L, Xin H, Suijin W, Shao D, Chengxin D, Lisi H, Zesheng L, Maohua Z, Lingji Z, Dongmei C, Yulian W, Xiaomei C, Suxia G, Weinkove R, Zhaoyang T, Chang H, Peng L, Xin D (2018) A novel generation 1928zT2 CAR T cells induce remission in extramedullary relapse of acute lymphoblastic leukemia. J Hemat Oncol 11(1):25
Gasser O, Sharples KJ, barrow C, Williams GM, Bauer E, Wood CE, Mester B, Dzhelali M, Caygill G, Jones J, Haman CM, Hinder VA, Macapagal J, McCuster M, Weinkove R, Painter GF, Brimble MA, Findlay MP, Dunbar PR, Hermans IF (2018) A phase I vaccination study with dendritic cells loaded with NY-ESO-1 & a-galactosylceramide: induction of polyfunctional T cells in high risk melanoma patients.Cancer Immunol Immunother 67(2):285-298
2017
Speir M, Authier-Hall A, Brooks CR, Farrand KJ, Compton BJ, Anderson RJ, Heiser A, Osmond TL, Tang CW, Berzofsky JA, Terabe M, Painter GF, Hermans IF, Weinkove R (2017) Glycolipid-peptide conjugate vaccines enhance CD8+ T cell responses against human viral proteins. Scientific Reports7:14273
Lucas N, Humble M, Sim D, Balm M, Carter JM and Weinkove R (2017) Temporal changes in neutropenic blood culture isolates: and disease associations: a single centre series of 1139 episodes. Int Med J 47(8):962-965
Speir, M., Hermans, I.F. and Weinkove, R. (2017) Engaging natural killer T cells as 'universal helpers' for vaccination. Drugs
2016
Collings S, Thompson O, Hirst E, Goossens L, George A, Weinkove R (2016) Non-Invasive Detection of Anaemia Using Digital Photographs of the Conjunctiva. PLoS One
